AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Co2 molecular geometry11/25/2023  
So, for the time being, put Carbon in the middle and circle it with four dots. 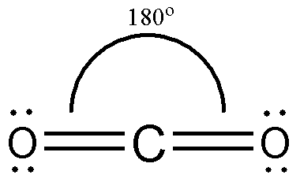
Let’s look at the valence electrons of all the atoms in the molecule to see how the bonds are formed and how they are organized. Carbon atoms share electrons with the two Oxygen atoms on the terminals and link to each other. The Carbon atom has the most prominent position in CO2 because it is the molecule’s least electronegative atom. Here is the Lewis dot diagram for CO2 to show you how it is drawn and how it is used. Understanding the arrangement of atoms and electrons involved in the bond formation is made easier by this structure. The molecule’s bonds are shown by lines drawn on the surface of the molecule. As a result of the Lewis dot structure, the valence electrons may be seen by placing small dots around the atoms. The valence shell electron configuration of a molecule is shown pictorially by the Lewis dot structure. Before learning about the Lewis dot diagram for CO2, it is necessary to have a firm grasp of what constitutes a Lewis structure. To better understand the arrangement of electrons in molecules and their form, this structure is useful. The paucity of vibrational structure in the TPES is discussed in terms of various fragmentation processes.Any molecule’s molecular geometry cannot be understood without first having a firm grasp of the Lewis structure. Results from the TPES indicate that the molecular geometry is distorted upon ionization, that production of BF occurs with low probability at threshold, and that autoionization may be responsible for the contrast between photoelectron spectra and the TPES. Separate mass analyzed efficiency curves measured near the appearance energies for BF and BF are also presented and discussed in terms of the thermochemical data which can thereby be derived along with the problems of assigning the precise appearance potential. The TPES, along with the total ionization efficiency curve obtained simultaneously, is presented for the first four electronic states of the BF ion. There is some indication for production of ions with vibrational excitation in the bending mode of X state. The spin-orbit splitting of the X state is readily observed. Production of ions with vibrational excitation in the symmetric stretching mode is observed for each state with the longest progression occurring for the A state. Autoionization is present in the vicinity of most of the X and A it states. Detailed threshold electron and total ionization spectra are presented for three electronic states (X, A, B) of the CO ion. The vibrational intensity distributions in the TPES of the A and B states compare more closely to the HeI spectrum. Results from the TPES indicate that the reported enhancement of the V(l) through V(4) levels of the X state in the NeI PES is due to autoionization. The V(0) level is the only member observed in the TPES for the X vibrational progression. Autoionization is prominent throughout the X and A states. The TPES and the total ion spectrum of the first three electronic states of N are illustrated and discussed. Photoionization efficiency curves are collected either as total ionization efficiency or as separate mass analyzed curves by employing a monopole mass filter. 014 eV for the TPES of the P state when using a. Electron energy discrimination is accomplished by using a collimated hole structure which operates on the principle of angular discrimination of electrons possessing more than thermal velocities. The instrument incorporates a 1 meter vacuum UV monochromator to disperse the light from the Hopfield continuum produced by a Velonex 360 high power pulse generator. The construction of an instrument capable of measuring the simultaneous photoion and threshold photoelectron spectra (TPES) is described in detail. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
